 |
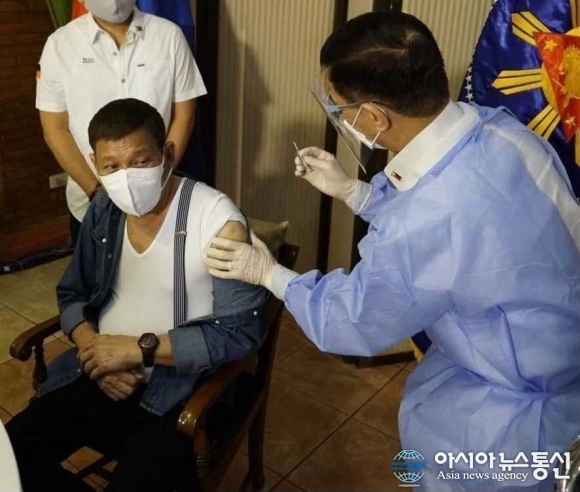
| This photo shows Health Sec. Francisco Duque III administering the Sinopharm vaccine to President Rodrigo Duterte on May 3, 2021 (Photo by Sen. Bong Go, Facebook) |
[Asia News Communication = Reporter Reakkana] MANILA — The country’s Food and Drug Administration said the COVID-19 vaccine administered to President Rodrigo Duterte was covered by the compassionate use permit granted to the chief executive’s security group, according to PhilStar. FDA Director-General Eric Domingo said this in an interview with ABS-CBN’s Teleradyo Tuesday after Duterte received his first shot of the jab developed by Sinopharm Group Co. Ltd.
The country’s FDA has yet to approve the vaccine for emergency use. The Chinese-made jab has been restricted to compassionate use only in the Philippines. In February, the FDA granted the Presidential Security Group’s application for a compassionate special permit to access 10,000 doses of the Sinopharm vaccine. A CSP allows the use of unregistered medical products, such as the Sinopharm jab, for limited off-label use.
“When you say compassionate special permit, it’s not an authorization given by the FDA. In this case, the head of the PSG hospital guarantees they studied the vaccine and they take full responsibility for it,” Domingo said. It is unclear why Duterte, a president, was covered by the permit given to his security team. But Domingo said the wives and partners of PSG personnel are also covered by the CSP.














